|
3/2/2024 0 Comments Electron capture equation
Let’s compare what we know about our emitted particle to the remaining answer choices. 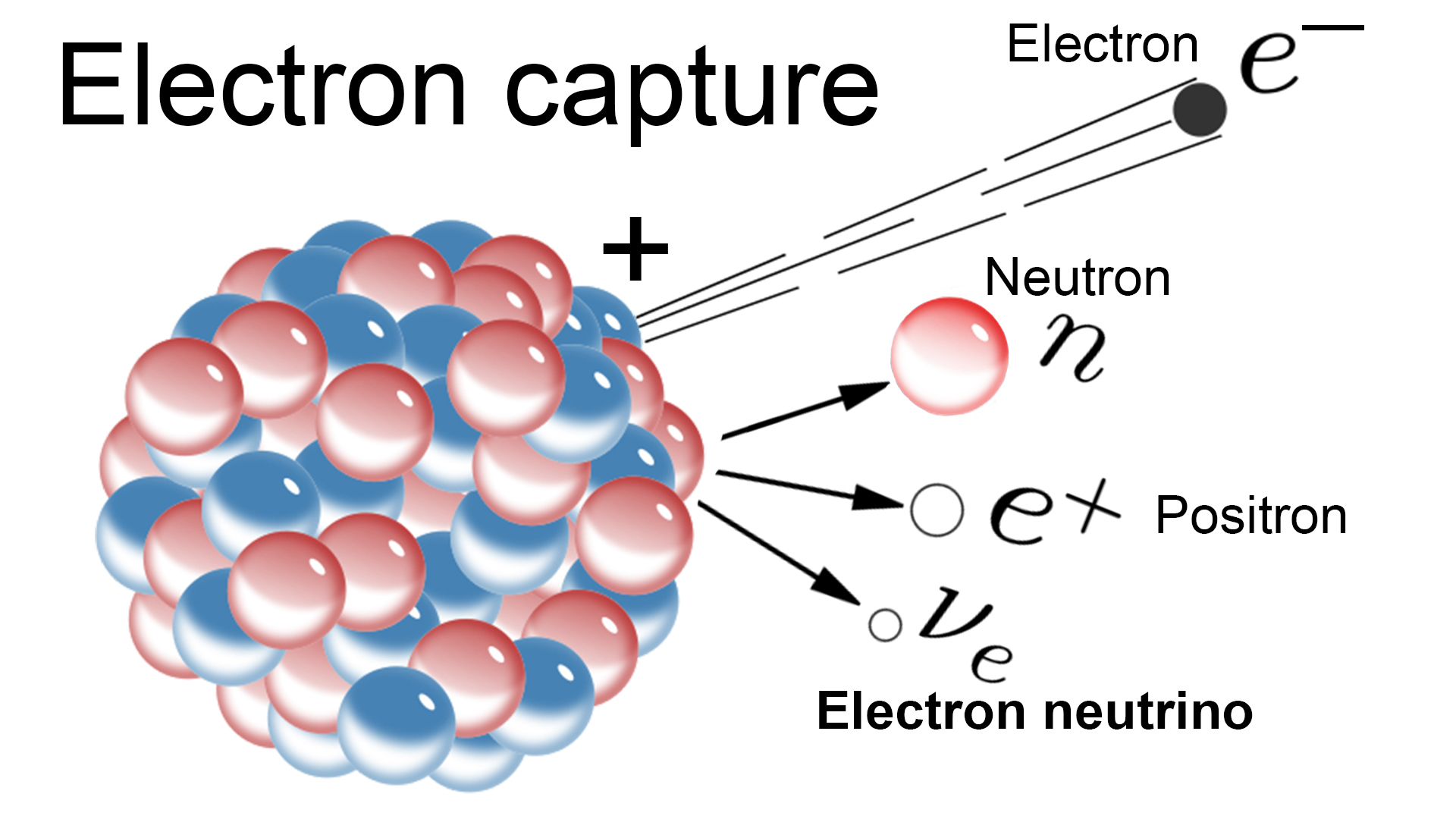
Our emitted particle’s symbol represents an electron with a mass number of zero and a charge of positive one. 
Let’s identify our emitted particle using nuclide notation, where X represents the symbol of the element or particle, where A represents the mass number, which for nuclei is the sum of the protons and neutrons, and where Z represents the charge, which for nuclei is the number of protons. Since the particle being emitted is expressed here as a product, we can eliminate answer choice (E). It is worth noting that electron capture is still considered radioactive decay, since gamma rays and X-rays are emitted during this process. Emission means that the distinguishing particle or radiation of the type of decay must be written as a product in the nuclear equation, while the final answer choice (E) uses the word capture, which means the particle, in this case an electron, is taken into the nucleus and must be written as a reactant in the nuclear equation. In the first four answer choices, we see that the decay process is described as emission. The given equation shows that boron-8 is decaying to form beryllium-8, and a particle or form of energy is also emitted. Radioactive decay is the process by which an unstable nucleus spontaneously emits particles or radiation. The question tells us that the equation given represents a decay process. Which decay process is represented by the following equation? (A) Positron emission, (B) beta emission, (C) alpha emission, (D) gamma emission, or (E) electron capture?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
